The Centers for Medicare & Medicaid Services (CMS) issued a proposed procedural notice outlining a new Medicare coverage pathway to achieve more timely and predictable access to new medical technologies for people with Medicare. The new Transitional Coverage for Emerging Technologies (TCET) pathway for Breakthrough Devices supports both improved patient care and innovation by providing a clear, transparent, and consistent coverage process while maintaining robust safeguards for the Medicare population.
The TCET pathway uses current national coverage determination (NCD) and coverage with evidence development (CED) processes to expedite Medicare coverage of certain Breakthrough Devices. TCET is voluntary and aims to reduce uncertainty about coverage options through a pre-market evaluation of potential harms and benefits of technologies while identifying any important evidence gaps. Additionally, the TCET pathway includes an evidence development framework that provides manufacturers with opportunities for increased pre-market engagement with CMS and, in an effort to reduce manufacturer burden, an unprecedented level of flexibility to address evidence gaps to support Medicare coverage. Specifically, TCET allows for any evidence gaps to be addressed through fit-for-purpose studies. A fit-for-purpose study is one where the study design, analysis plan, and study data are appropriate for the question the study aims to answer. Further, the TCET pathway will help coordinate benefit category determination, coding, and payment reviews.
When developing the pathway, CMS solicited extensive feedback from patient groups, medical professionals, device manufacturers, innovators, and other Federal agencies. This feedback included requests for CMS to utilize a more agile, iterative evidence review process that considers fit-for-purpose study designs, including those that make secondary use of real-world data. CMS partnered with the Agency for Healthcare Research and Quality (AHRQ) to develop a comprehensive approach that incorporates greater flexibility into the proposed CED paradigm and allows fit-for-purpose study designs. CMS will continue to engage with these stakeholders to ensure that Medicare promotes access to emerging medical technologies while maintaining appropriate safeguards and rigorous evidence standards essential to the health of Medicare beneficiaries.
The public will have 60 days to provide comment on the procedural notice. CMS will respond to public comments in a subsequent final notice.
Background
Today’s announcement fulfills CMS’ commitment to release a new Medicare coverage pathway uniquely suited to new, innovative technologies that better balances the needs of all stakeholders, particularly beneficiaries. As part of our commitment to fostering innovation and ensuring patient-centered care, CMS created the TCET pathway to provide a mechanism for coverage for certain new, innovative technologies with limited or developing evidence in the Medicare population using a transparent and predictable evidence generation framework that, when appropriate, not only develops reliable evidence for patients and their physicians to make health care decisions but also provides safeguards to ensure that Medicare beneficiaries are protected and continue to receive high-quality care.
TCET Pathway
The TCET pathway is intended to balance multiple considerations when making coverage determinations: (1) facilitating early, predictable, and safe beneficiary access to new technologies; (2) reducing manufacturers’ and innovators’ uncertainty about coverage by evaluating early the potential benefits and harms of technologies; and (3) encouraging evidence development if evidence gaps exist.
TCET Pathway at a Glance
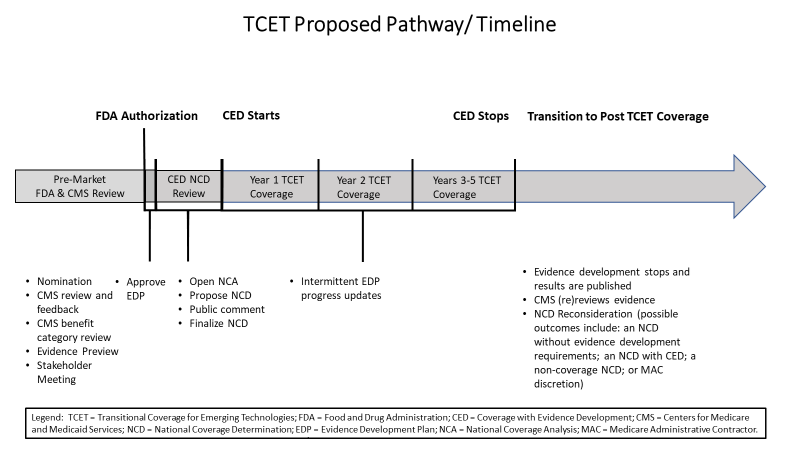
Evidence Development Under TCET
It is important that any new coverage pathway for emerging technologies facilitate evidence development when evidence gaps exist to ensure that Medicare beneficiaries have access to new technologies that will improve health outcomes. In instances where evidence gaps exist, CMS is committed to working closely with manufacturers to develop evidence to facilitate coverage under the TCET pathway. Specifically, CMS will work with manufacturers to ensure that evidence development under the TCET pathway does not require duplicative or conflicting evidence development with any FDA post-market requirements for the device.
Evidence development to support Medicare coverage through the TCET pathway will be based upon a more transparent and predictable evidence-generation framework. In addition to the TCET procedural notice, CMS issued newly proposed criteria in an updated CED guidance document based on the November 2022 AHRQ Report [https://effectivehealthcare.ahrq.gov/products/coverage-evidence-development/research-report], and February 2023 MEDCAC [https://www.cms.gov/medicare-coverage-database/view/medcac-meeting.aspx?medcacid=79], as well as National Coverage Analysis Evidence Review guidance to allow for fit-for-purpose study designs more clearly. We expect to publish more detailed fit-for-purpose guidance later this year. Additionally, CMS published the first in a series of guidance documents that review health outcomes and their clinically meaningful differences within priority therapeutic areas. The public will have an opportunity to provide comment on these guidance documents. CMS’ guidance documents can be accessed here: https://www.cms.gov/medicare-coverage-database/reports/national-coverage-medicare-coverage-documents-report.aspx?docTypeId=1#. Finally, CMS will soon announce the opening of an NCD pilot that will incorporate aspects of the new evidence development framework outlined in the TCET procedural notice and guidance documents.
Key components of the TCET pathway to facilitate evidence development include:
An Evidence Preview, which is a focused literature review that would provide early feedback on the strengths and weaknesses of the available evidence, including any evidence gaps, for a specific item or service. It is intended to inform judgments by CMS and manufacturers about the best available coverage options for an item or service and offers greater efficiency, predictability, and transparency to manufacturers and CMS on the state of the evidence and any notable evidence gaps. CMS intends for Evidence Previews to be conducted by a contractor using standardized evidence grading, risk of bias assessment, and applicability assessment according to a protocol initially developed in collaboration with the AHRQ in 2020, which is reflected in the proposed Evidence Review guidance document. An Evidence Preview will be made publicly available on the CMS website when a tracking sheet is posted announcing the opening of an NCD.
An Evidence Development Plan (EDP), which will be developed by the manufacturer to address any evidence gaps identified in the Evidence Preview. EDPs may include traditional clinical study designs and/or fit-for-purpose study designs, including those that rely on secondary use of real-world data, provided those study designs follow all applicable CMS guidance documents. The development of an EDP will include CMS-AHRQ collaboration to evaluate the EDP to ensure that it meets established standards of scientific integrity and relevance to the Medicare population. Additionally, it will include CMS engagement with the manufacturer to provide feedback and discuss any recommended refinements. Elements of the EDP, specifically the non-proprietary information, will be made publicly available on the CMS website when a proposed NCD is posted.
Device Eligibility
Given the unique FDA criteria for Breakthrough designation status, the TCET pathway will apply to certain eligible FDA-designated Breakthrough Devices because this is the area with the most immediate need for a pathway like TCET.
Appropriate candidates for the TCET pathway would include those devices that are:
- Certain FDA-designated Breakthrough Devices;
- Determined to be within a Medicare benefit category;
- Not already the subject of an existing Medicare NCD; and
- Not otherwise excluded from coverage through law or regulation.
In section 201(h)(1) of the Federal Food, Drug, and Cosmetic Act (21 U.S.C. 321(h)(1), the definition of device includes diagnostic laboratory tests. Diagnostic lab tests are a highly specific area of coverage policy development, and CMS has historically delegated review of many of these tests to specialized Medicare Administrative Contractors (MACs). CMS believes that the majority of coverage determinations for diagnostic tests granted Breakthrough designation status should continue to be determined by the MACs through existing pathways.
Nominations for the TCET Pathway
Manufacturers of FDA-designated Breakthrough Devices may self-nominate to participate in the TCET pathway on a voluntary basis. If interested in the TCET pathway, manufacturers simply need to notify CMS of their interest in TCET via an email box that would be established specifically for this purpose. The appropriate timeframe for manufacturers to submit TCET pathway nominations to CMS is approximately 12 months prior to the anticipated FDA decision on a submission as determined by the manufacturer. The notice outlines the information that manufacturers should include in the self-nomination packet. CMS’ consideration of nominations will include a meeting with the manufacturer to discuss the technology and answer any questions on the process. CMS will meet with FDA to learn more information about the technology and receive preliminary information on the device and potential review timing. As part of the nomination consideration process, the technology may undergo a benefit category review. Upon completion of CMS’ review of the nomination, CMS will notify the manufacturer by email whether the product is an appropriate candidate for the TCET pathway.
TCET and the NCD process
If a device that is accepted into the TCET pathway receives FDA marketing authorization, CMS will initiate the NCD process by posting a tracking sheet following FDA market authorization (that is, the date the technology receives premarket approval (PMA); 510(k) clearance; or the granting of a De Novo request) pending a CMS and AHRQ-approved EDP (in cases where there are evidence gaps as identified in the Evidence Preview). The process for Medicare coverage under the TCET pathway would follow the NCD statutory timeframes in section 1862(l) of the Social Security Act. CMS’ goal is to finalize a TCET NCD within six months after FDA market authorization.
Duration of Coverage Under the TCET Pathway
Coverage under the TCET NCD will continue only as long as needed to facilitate the timely generation of evidence that can inform patient and clinician decision making. The duration of transitional coverage through the TCET pathway will be tied to the CMS- and AHRQ-approved EDP. The review date specified in the EDP will provide one additional year of coverage after study completion to allow manufacturers to complete their analysis, draft one or more reports, and submit them for peer-reviewed publication. In general, we anticipate this period would last for three to five years as evidence is generated to address evidence gaps identified in the Evidence Preview and lead to a predictable, long-term Medicare coverage determination.
Transition to Post-TCET Coverage
CMS intends to conduct an updated evidence review within six calendar months of the review date specified in the EDP. Similar to the process used for the Evidence Preview, CMS intends to engage a third-party contractor to conduct the systematic literature review. The contractor will perform a qualitative evidence synthesis and compare those findings against the benchmarks for each outcome specified in the original NCD. CMS will conduct quality assurance on the contractor review. CMS will also review applicable practice guidelines and consensus statements and consider whether the conditions of coverage remain appropriate.
Based upon this assessment, when appropriate, CMS will open an NCD reconsideration by posting a proposed decision that proposes one of the following outcomes: (1) an NCD without evidence development requirements; (2) an NCD with continued evidence development requirements; (3) a non- coverage NCD; or (4) rescinding the NCD, resulting in coverage decisions being made by MACs under section 1862(a)(1)(A) of the Social Security Act. Standard NCD processes and timelines will continue to apply, and following a 30-day public comment period, CMS will have 60 days to finalize the NCD reconsideration.
Coverage of Similar Devices
FDA market-authorized Breakthrough Devices are often followed by similar devices that other manufacturers develop. CMS believes that it is important to let physicians and their patients make decisions about the best available treatment depending on the patient’s individual situation. CMS is seeking public comment on whether similar devices to the Breakthrough Device should be addressed under a separate NCD or should be subject to the same coverage conditions as the Breakthrough Device, including a requirement to propose an EDP.
For more information on the TCET pathway, please visit: https://www.federalregister.gov/public-inspection/2023-13544/medicare-program-transitional-coverage-for-emerging-technologies
###