Previous Spotlights
Learn more about our earlier work. Visit the Spotlight page to learn about our most recent accomplishments
On July 20, 2022, the Department of Justice announced criminal charges against 36 defendants in 13 federal districts across the United States for more than $1.2 billion in alleged fraudulent telemedicine, cardiovascular and cancer genetic testing, and durable medical equipment (DME) schemes.
The nationwide coordinated law enforcement action includes criminal charges against a telemedicine company executive, owners and executives of clinical laboratories, durable medical equipment companies, marketing organizations, and medical professionals. In connection with the enforcement action, the department seized over $8 million in cash, as well as luxury vehicles and other fraud proceeds.
Additionally, the Centers for Medicare & Medicaid Services (CMS), Center for Program Integrity (CPI) announced today that it took adverse administrative actions against 52 providers involved in similar schemes.
“The Centers for Medicare & Medicaid Services continues to aggressively investigate fraud, waste and abuse and has taken action to protect patients, critical health care resources and to prevent losses to the Medicare Trust Fund,” said CMS Administrator Chiquita Brooks-LaSure. “Work like this to combat fraud, waste, and abuse in our federal programs would not be possible without the successful partnership of CMS, the Department of Justice, and the U.S. Department of Health and Human Services Office of Inspector General.”

The Healthcare Fraud Prevention Partnership (HFPP) has released its latest white paper, “Exploring Fraud, Waste, and Abuse Within Telehealth (PDF)," in response to the evolving fraud, waste, and abuse associated with the delivery of care conducted through telehealth services. This resource was developed in collaboration with Stanford University School of Medicine and includes direct input from HFPP Partners – representing federal and state agencies, private payers, law enforcement organizations, and healthcare anti-fraud associations.
The white paper provides background on the use of telehealth services prior to and during the COVID-19 Public Health Emergency, along with descriptions of newly developed and repurposed fraud schemes related to telehealth, including: billing for medically unnecessary services, upcoding, improbable days, and miscoding virtual COVID-19 related care.
Additionally, HFPP Partners, industry experts, and researchers offer numerous strategies and lessons learned to help readers anticipate potential telehealth fraud and mitigate vulnerabilities.
For more information, please see:
The Healthcare Fraud Prevention Partnership (HFPP) celebrated its 10th year as a leader in strengthening the nation’s fight against healthcare fraud, waste, and abuse. The HFPP is a voluntary public-private partnership that helps detect and prevent healthcare fraud through data and information sharing. Partners include federal government, state agencies, law enforcement, private health insurance plans, employer organizations, and healthcare anti-fraud associations.
The Partnership's purpose then and now is to exchange facts and information between the public and private sectors and enable the performance of sophisticated data analytics against our unique cross-payer data set to reduce the prevalence of healthcare fraud. Read the press release from when the HFPP was announced.
The HFPP has come a long way since its inception in 2012. Check out some of the key accomplishments:
- In 2012, when the HFPP was formed, it consisted of 21 Partners. Driven by the importance and value of the HFPP’s mission, membership has grown over the last 10 years by over 250 members.
- The HFPP first began with professional claim types only, then expanded in 2019 to include institutional; in 2021, pharmacy claims were added for more comprehensive analytic insights.
- Starting initially with original claims data, the Partnership is now conducting its analysis against adjusted claims to detect industry-wide fraud schemes more precisely.
Over the next decade and beyond, the HFPP looks forward to continuing its work with Partners to enhance fraud-fighting efforts through innovative initiatives, enhanced study analytics, enriched infrastructure, expanded engagement, increased membership, and strengthened strategic partnerships.
The Healthcare Fraud Prevention Partnership (HFPP) released its newest White Paper, “Fraud, Waste, and Abuse in the Context of COVID-19 (PDF)” in collaboration with Stanford University School of Medicine.
This new HFPP white paper provides a background on COVID-19 and its impact on healthcare delivery. The paper then highlights trending fraud schemes and offers strategies for healthcare payers to consider and apply.
Broadly, this white paper outlines important steps that federal and state agencies, private payers, and law enforcement have taken in identifying and responding to fraud, waste, and abuse related to the delivery of care for COVID-19. These actions and lessons learned may allow these parties to anticipate vulnerabilities moving forward, providing a foundation for navigating a changed healthcare landscape and future healthcare challenges.
For additional information, please see:
On April 20, 2022, the Department of Justice announced criminal charges against 21 defendants in nine federal districts across the United States for their alleged participation in various health care related fraud schemes that exploited the COVID-19 pandemic. These cases allegedly resulted in over $149 million in COVID-19-related false billings to federal programs and theft from federally-funded pandemic assistance programs. This announcement builds on the success of the DOJ’s May 2021 COVID-19 Enforcement Action and involves the prosecution of various COVID-19 health care fraud schemes.
Additionally, the Center for Program Integrity, Centers for Medicare & Medicaid Services (CPI/CMS) separately announced today that it has taken an additional 28 administrative actions against providers for their alleged involvement in fraud, waste and abuse schemes related to the delivery of care for COVID-19, as well as schemes that capitalize upon the Public Health Emergency.
“We are committed to working closely with our law enforcement partners to combat fraud, waste and abuse in our federal health care programs,” said CMS Administrator Chiquita Brooks-LaSure. “The administrative actions CMS has taken protect the Medicare Trust Funds while also safeguarding people enrolled in Medicare.”
On May 4, 2022, the Department of Justice, together with federal and state law enforcement partners, today announced criminal charges against 14 defendants in eight federal districts across the United States for their alleged involvement in crimes related to the unlawful distribution of opioids. Twelve of the defendants were licensed medical professionals at the time of these alleged offenses.
Additionally, the Centers for Medicare & Medicaid Services’ (CMS) Center for Program Integrity has taken six administrative actions against providers for their alleged involvement in these offenses.
"Patient care and safety are top priorities for us, and CMS has taken administrative action against eight providers to protect critical resources entrusted to Medicare while also safeguarding people with Medicare,” said CMS Administrator Chiquita Brooks-LaSure. "These actions to combat fraud, waste, and abuse in our federal programs would not be possible without the close and successful partnership of the Centers for Medicare & Medicaid Services, the Department of Justice, and the U.S. Department of Health and Human Services Office of Inspector General."
Open Payments released a short video to show how Open Payments works, what’s in the data, and how to use the search tool.
Open Payments is a national disclosure program that promotes a transparent and accountable health care system by making the financial relationships between the health care industry and health care providers available to the public. As of March 2021, the Open Payments data site houses more than 76 million records accounting for $53.01 billion of payments or transfers of value.
Check out the video to learn more!
On October 14, 2021, Acting United States Attorney Jennifer Arbittier Williams of the Eastern District of Pennsylvania announced three settlements and the filing of a complaint under the False Claims Act. These are the latest law enforcement actions in the national investigation into the scheme of improper billing involving P-Stim electro-acupuncture devices. Federal healthcare programs do not reimburse for P-Stim devices, whether they are characterized as an electro-acupuncture device or as an implantable neuro-stimulator.
The four enforcement actions announced by U.S. Attorney Williams allege that certain parties sold P-Stim devices and/or promoted them as billable to Medicare and other federal healthcare programs, which then caused providers to submit fraudulent claims. The United States alleges that these promoters profited by conspiring together to make false representations to providers that P-Stim was reimbursable under billing codes that paid thousands of dollars per procedure. Those codes were meant for legitimate, surgically implanted neuro-stimulators to manage chronic pain. However, P-Stim devices can be applied in a few minutes in an office setting without anesthesia by someone with minimal training. The promoters allegedly had knowledge that the P-Stim devices were not reimbursable by federal healthcare programs but pushed the non-surgical devices anyway.
“CMS is dedicated to removing fraudulent actors and protecting the people who rely on our programs,” said CMS Administrator Chiquita Brooks-LaSure. “We thank our partners at the Department of Justice and Department of Health and Human Services Office of Inspector General for collaborating with us to identify, investigate, and eliminate waste, fraud, and abuse in our federal health care programs.”
CMS aggressively investigates providers and suppliers to ensure appropriate payments are billed to federal healthcare programs, including through the use of data-analytic investigative tools. The U.S. Attorney’s Office for the Eastern District of Pennsylvania and other law enforcement partners have pursued and settled various False Claims Act cases against P-Stim providers and suppliers, recovering millions. Including the three settlements, the national P-Stim initiative has so far produced over 15 False Claims Act settlements across the country worth approximately $15 million. Additionally, outside of DOJ litigation, there are administrative enforcement actions by other federal agencies as well. In particular, CMS, through its Program Integrity Contractors, is auditing and recovering improperly paid P-Stim claims. HHS-OIG is also pursuing civil money penalties and exclusion remedies.
On Sept. 17, 2021, the Department of Justice announced criminal charges against 138 defendants in 31 federal districts across the U.S. The defendants were doctors, nurses, and other licensed medical professionals who allegedly participated in health care fraud schemes resulting in more than $1.4 billion in losses.
The charges include approximately $1.1 billion in telemedicine fraud, $29 million in COVID-19 health care fraud, $133 million in fraud arising from substance abuse treatment facilities (“sober homes”), and $160 million from illegal opioid distribution and other health care fraud schemes across the nation.
“Every dollar saved is critical to the sustainability of our Medicare programs and meeting the needs of seniors and people with disabilities,” said Chiquita Brooks-LaSure, Centers for Medicare & Medicaid Services (CMS) administrator. “CMS has taken actions against 28 providers on behalf of people with Medicare coverage and to protect the Medicare Trust Fund. Actions like this to combat fraud, waste, and abuse in our federal programs would not be possible without the successful partnership of the Centers for Medicare & Medicaid Services, the Department of Justice, and the U.S. Department of Health and Human Services Office of Inspector General.”
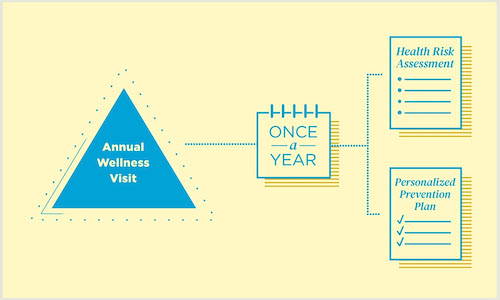
This video was created to emphasize the differences between an Initial Preventive Physical Examination (IPPE), Routine Physical Exam (RPE), and Annual Wellness Visit (AWV). The video provides health care professionals with guidance to understand expectations and requirements when submitting documentation for Annual Wellness Visits (AWV) for Medicare beneficiaries.
For more information about health risk assessments, coding, diagnosis, billing, and initial and subsequent components of an Annual Wellness Visit, visit the MLN Matters booklet Annual Wellness Visit, on the CMS website. This article can be found at MLN Matters® booklet (PDF).
Open Payments is a national disclosure program that promotes transparency in the health care industry by making information about the financial relationships between pharmaceutical companies and medical device manufacturers (reporting entities) and physicians and teaching hospitals (covered recipients) available to the public.
Starting in 2021, the type of data collected for Open Payments will expand to include:
- Five new provider types (in addition to physicians and teaching hospitals): physician assistants, nurse practitioners, clinical nurse specialists, certified registered nurse anesthetists and anesthesiologist assistants, and certified nurse midwives.
- Three new categories for describing the nature of payments: debt forgiveness, long-term medical supply or device loan, and acquisitions.
- Reporting requirements for the ‘device identifier’ component of the unique device identifier for medical supplies and devices.
The key dates to be aware of for these changes are:
- January 2021: Reporting entities begin collecting data that reflects these changes, including information on certain transactions between reporting entities and the newly-added covered recipient groups. Data collection continues for the duration of the year.
- February and March 2022: Reporting entities submit data from calendar year 2021 to Open Payments.
- April to mid-May 2022: Covered recipients, including the newly-added covered recipients, have the opportunity to review data reported about them before it is published and dispute it if necessary.
To prepare, learn more details about these changes for reporting entities and covered recipients and sign up for updates on the Contact Us page.
The Center for Program Integrity, Centers of Medicare & Medicaid Services (CMS) is proud of the Department of Justice’s announcement of one of the largest coordinate law enforcement actions to combat health care fraud related to COVID-19. On May 26, 2021, the Department of Justice announced criminal charges against 14 defendants. Additionally, CMS separately took adverse administrative actions against over 50 providers for their involvement in health care fraud schemes relating to COVID-19 or their abuse of CMS programs that were designed to encourage access to medical care during the pandemic.
Every dollar saved is critical to the sustainability of our Medicare program and meeting the needs of our beneficiaries. Actions like this to combat fraud, waste and abuse in our federal programs would not be possible without the successful partnership of CMS, the Department of Justice and Department of Health and Human Services Office of Inspector General.
Find more information please see the following links:
- Read the DOJ press release.
- Read - OIG Media Materials webpage.
- Visit the OIG COVID-19 Portal.
If you believe you have been the victim of COVID-19 fraud, immediately report it to:
- CMS/Medicare Hotline 1-800-Medicare
- HHS-OIG Hotline 1-800 HHS-TIPs
Stay Up to Date with CPI
Connect with CPI as we host or attend various events throughout the year, join our mailing list to stay informed on Program Integrity news, or find the most appropriate vehicle to report suspected fraud, waste, or abuse.